Abstract
Background: Mantle cell lymphoma (MCL), an aggressive B-cell non-Hodgkin lymphoma, remains incurable with standard therapies. The highly selective, potent Bruton tyrosine kinase (BTK) inhibitor acalabrutinib was approved by the US Food and Drug Administration for the treatment of relapsed/refractory MCL based on clinical data showing a high rate of durable responses and a favorable safety profile (Lancet 2017;391:659-667). Here, we present long-term follow-up in these patients.
Methods: Eligible patients were aged ≥18 years, had confirmed MCL, Eastern Cooperative Oncology Group performance status ≤2, and had relapsed and/or were refractory to 1-5 prior therapies. Exclusion criteria included prior BTK or BCL-2 inhibitor exposure and concomitant warfarin or equivalent vitamin K antagonists. Oral acalabrutinib 100 mg twice daily was administered until progressive disease or unacceptable toxicity. Response was assessed by investigators based on the Lugano classification (J Clin Oncol 2014;32:3059-3068).Analysis of minimal residual disease using next-generation sequencing (10-6) is ongoing for a subset of patients with available samples and will be presented upon completion.
Results: A total of 124 patients were treated; 80% were men, and median age was 68 years (range, 42-90 years) with 65% aged ≥65 years. At baseline, 93% of patients had Eastern Cooperative Oncology Group performance status ≤1, 8% had bulky lymph nodes ≥10 cm, 72% had extranodal involvement, and 44%/17% had intermediate-/high-risk simplified MCL International Prognostic Index scores. The median number of prior therapies was 2 (range, 1-5); 24% were refractory to the most recent prior treatment.
As of February 12, 2018, median time on study was 26.3 months (range, 0.3-35.1 months), and 40% of patients remain on treatment. Median relative dose intensity (ratio of actual to planned cumulative dose during drug exposure period) was 99% (range, 27%-100%). Investigator-assessed overall response rate was 81% (95% CI: 73%, 87%), with 43% (95% CI: 34%, 52%) achieving complete response (Table). Overall response rates were consistent across prespecified subgroups of tumor bulk, presence of refractory disease and number/type of prior treatment.
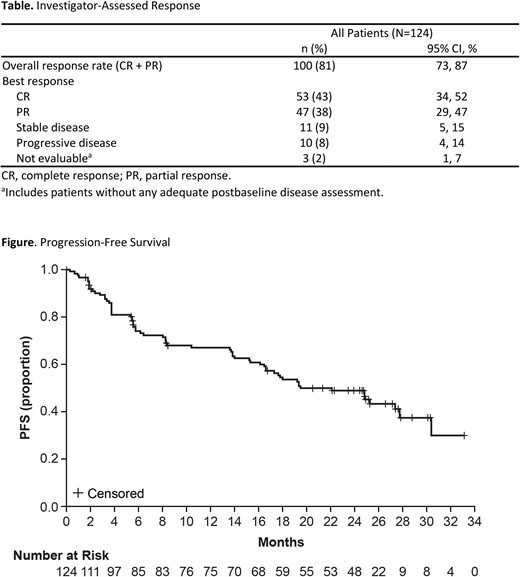
Median duration of response was 25.7 months (95% CI: 17.5 months, not reached). Median progression-free survival (PFS) was 19.5 months (95% CI: 16.5 months, 27.7; Figure). Median overall survival (OS) was not reached; the estimated 24-month OS rate was 72% (95% CI: 64%, 80%).
The most frequent adverse events (AEs; ≥20%) were primarily Grade 1/2 and included headache (38%), diarrhea (36%), fatigue (28%), cough (22%) and myalgia (21%). Grade 3/4 AEs (≥5%) included anemia (10%), neutropenia (10%) and pneumonia (6%). There were 13 patients (10%) with 16 cardiac events, including 4 Grade 3/4 events (3%) in 1 patient each (acute coronary syndrome, acute myocardial infarction, cardiorespiratory arrest, coronary artery disease). Four patients had hypertension events (3%); 1 event was Grade 3. The most common bleeding events were contusion (13%) and petechiae (9%); all bleeding events were Grade 1/2 except for 3 Grade 3 events (gastrointestinal hemorrhage, hematuria, hematoma). Grade 3/4 infections occurred in 15% of patients and none were Grade 5; there was one case of cytomegalovirus viremia and one case of pneumocystis jiroveci pneumonia (both Grade 2).
Treatment discontinuation was primarily due to progressive disease (n=54; 44%) and AEs (n=10; 8%).Twelve AEs led to discontinuation in 10 patients; all of these AEs occurred in only 1 patient each.There were 43 deaths (35%), most commonly from progressive disease (n=29; 23%) or AEs (n=6; 5%). Deaths due to AEs included bilateral pulmonary embolism, critical aortic stenosis, myelodysplastic syndrome, pneumonia, suicide, and non-small cell lung cancer; none were considered to be related to acalabrutinib.
Conclusion: Response to acalabrutinib remained consistent during long-term (>24-month) follow-up, including high response rates, median PFS of 19.5 months, and a median OS that has not yet been reached, confirming efficacy in patients with relapsed/refractory MCL. The AE profile was largely similar to earlier reporting, with limited additional safety events observed with an additional year of follow-up.
Wang:Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Dava Oncology: Honoraria; AstraZeneca: Consultancy, Research Funding; Pharmacyclics: Honoraria, Research Funding; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Juno: Research Funding; MoreHealth: Consultancy; Acerta Pharma: Honoraria, Research Funding; Kite Pharma: Research Funding; Novartis: Research Funding. Rule:Celltrion: Membership on an entity's Board of Directors or advisory committees; Gilead Sciences, Inc.: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees; AstraZeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees; Janssen: Consultancy, Honoraria, Research Funding, Speakers Bureau; Roche: Honoraria; Kite: Membership on an entity's Board of Directors or advisory committees. Zinzani:Roche: Honoraria, Membership on an entity's Board of Directors or advisory committees; Merck: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Verastem: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; TG Pharmaceuticals: Honoraria, Membership on an entity's Board of Directors or advisory committees; PFIZER: Honoraria, Membership on an entity's Board of Directors or advisory committees; Astra Zeneca: Speakers Bureau; Takeda: Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria, Speakers Bureau; SERVIER: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Gilead: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celltrion: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Bayer: Membership on an entity's Board of Directors or advisory committees; TG Pharmaceuticals: Honoraria, Membership on an entity's Board of Directors or advisory committees; Bayer: Membership on an entity's Board of Directors or advisory committees; PFIZER: Honoraria, Membership on an entity's Board of Directors or advisory committees; Merck: Honoraria, Membership on an entity's Board of Directors or advisory committees; MSD: Honoraria, Speakers Bureau. Casasnovas:MSD: Honoraria; Merck: Honoraria; Takeda: Honoraria; Roche: Honoraria; Gilead Sciences: Research Funding; Roche: Research Funding; Janssen: Consultancy; Gilead Sciences: Consultancy; MSD: Consultancy; merck: Consultancy; takeda: Consultancy; Roche: Consultancy; Gilead Sciences: Honoraria; Janssen: Honoraria; Celgene: Honoraria. Smith:Genentech: Research Funding; Portola: Research Funding; Pharmacyclics: Research Funding; Merck Sharpe Dohme and Corp: Consultancy, Research Funding; Seattle Genetics: Research Funding; Acerta Pharma BV: Research Funding. Morschhauser:BMS: Membership on an entity's Board of Directors or advisory committees; Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees; Janssen: Other: Scientific Lectures; Epizyme: Consultancy; Roche: Membership on an entity's Board of Directors or advisory committees; Gilead: Consultancy, Membership on an entity's Board of Directors or advisory committees. Panizo:Janssen: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celgene: Speakers Bureau; BMS: Membership on an entity's Board of Directors or advisory committees; Acerta Pharma: Research Funding; Roche: Consultancy, Speakers Bureau. Davies:Janssen: Consultancy, Honoraria; Karyopharma: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Gilead: Honoraria, Research Funding; Acerta Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; F. Hoffman-La Roche: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Pfizer: Research Funding; Takeda: Consultancy, Membership on an entity's Board of Directors or advisory committees; Kite: Consultancy; ADC Therapeutics: Research Funding; Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; GSK: Research Funding. Jacobsen:Seattle Genetics: Consultancy; AstraZeneca: Consultancy; Merck: Consultancy. Kater:Janssen: Honoraria, Research Funding; Abbvie: Honoraria, Research Funding; Acerta/AZ: Research Funding; Genentech: Honoraria, Research Funding. Robak:Gilead: Consultancy; Janssen: Consultancy, Honoraria; AbbVie, Inc: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Roche: Consultancy, Honoraria. Dua:Acerta Pharma: Employment. Frigault:AstraZeneca: Employment, Equity Ownership. Izumi:Acerta Pharma: Employment, Equity Ownership, Patents & Royalties: Acerta Pharma, various patents for ACP-196. Nguyen:Acerta Pharma: Employment. Patel:Acerta Pharma: Employment, Equity Ownership. Yin:Acerta Pharma: Employment. Jurczak:European Medicines Agency: Consultancy; Astra Zeneca/Acerta: Consultancy, Research Funding; Sandoz-Novartis: Consultancy; Janssen: Consultancy, Research Funding; Gilead: Consultancy, Research Funding; Afimed: Research Funding; Bayer: Research Funding; Beigene: Research Funding; Celgene: Research Funding; Epizyme: Research Funding; Nordic Nanovector: Research Funding; Merck: Research Funding; Morphosys: Research Funding; Pharmacyclics: Research Funding; Servier: Research Funding; Roche: Research Funding; TG Therapeutics: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal